
PCR without the complexity.
The new standard in PCR performance.

True quantification
Direct molecular counting enables precise measurements with consistently low CVs

Superior sensitivity
Resolve alleles down to 0.004% MAF while maintaining linear performance across a wide dynamic range

Effortless multiplexing
Rapidly develop multiplex assays, saving valuable time and resources

Seamless compatibility
Use existing probes, no assay redesign needed
Applications
See how Countable PCR redefines what’s possible across genomics applications.
Application Note: Rapid, quantitative viral genome integrity measurements using multiplex Countable PCR assays
Workflow
A workflow simple enough to be adopted by any lab.
01. Prepare
Prepare reaction
Load sample and reaction components to spin column.
02. Generate Matrix
Generate matrix
Centrifuge to isolate single molecules.
03. Amplify
Amplify targets
Perform PCR to amplify target signal.
04. Count
Count targets
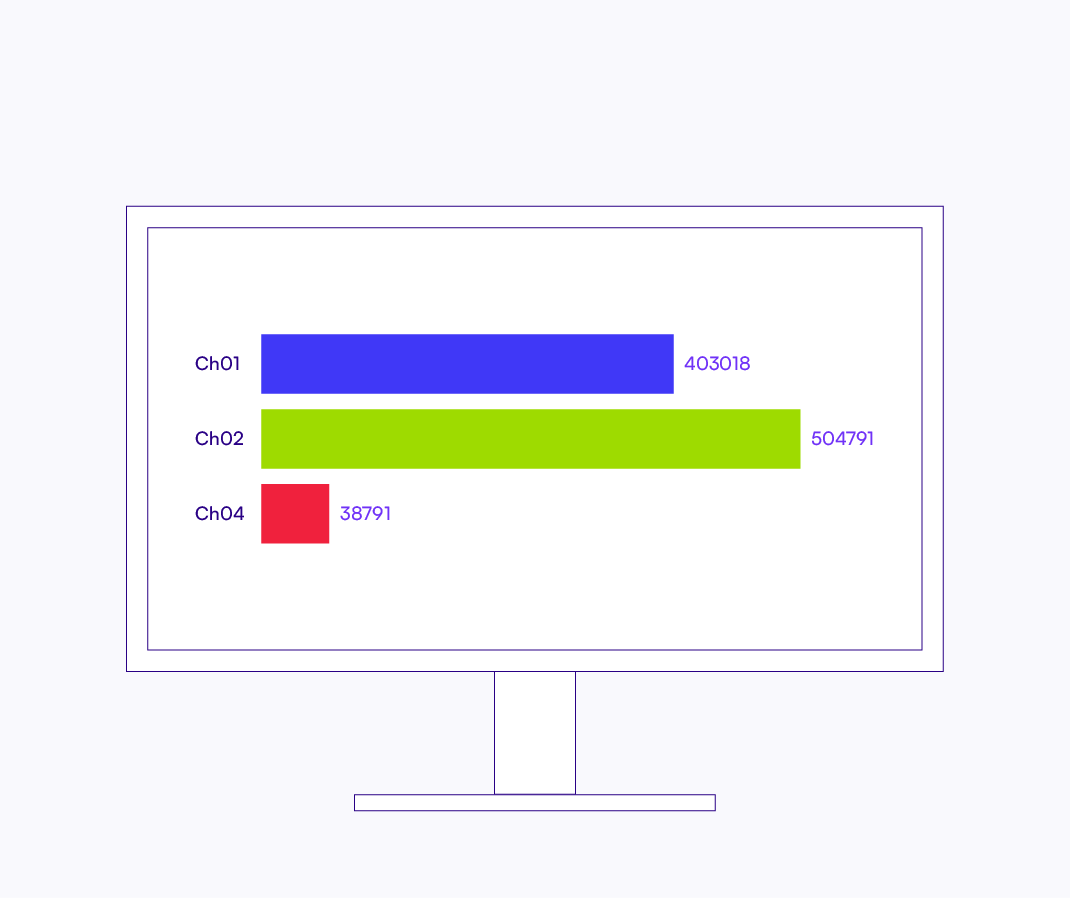
Quantify using 3D light sheet imaging.
Compatible sample types
Cell free DNA
Tissue DNA
FFPE DNA
cDNA
Plasmids
Nucleosomal DNA
Genomic DNA, sheared
Genomic DNA, undigested
Crude cell lysate
Virus with capsids

Key specifications
The performance is Countable.
<1% CVs
High reproducibility
Up to 10 targets*
Multiplexing per reaction
1- 96 samples
per run
1-35 µL
sample input
up to 5kb
amplicon length
Up to 1 million
Target molecules counted
* Available on Countable 10. Contact us for service projects today.
Workflow
Getting started is simple.
Find out how Countable PCR transforms your work.
Frequently Asked Questions
Yes - we have Pre-designed Assays that include expert-verified protocols and performance data for commonly tested targets. We are continually adding new assays to our menu.
It is simple to transition existing hydrolysis probe-based assays to Countable PCR - your existing probes and primers can be used directly, although we do recommend including a probe additive to reduce background signal. Learn more here.
Yes. The software automatically detects and counts target molecules from the 3D light sheet images for every sample and target. In addition, every run produces a QC report in a 21 CFR Part 11–compliant format, supporting GMP-ready traceability.
It will depend on which Countable System is used for analysis. You can see which dyes are supported on the instrument pages for Countable 4 and Countable 10.






