A Deep Sampling and Single Molecule Digital Counting Assay for Rare Molecule Detection
Abstract
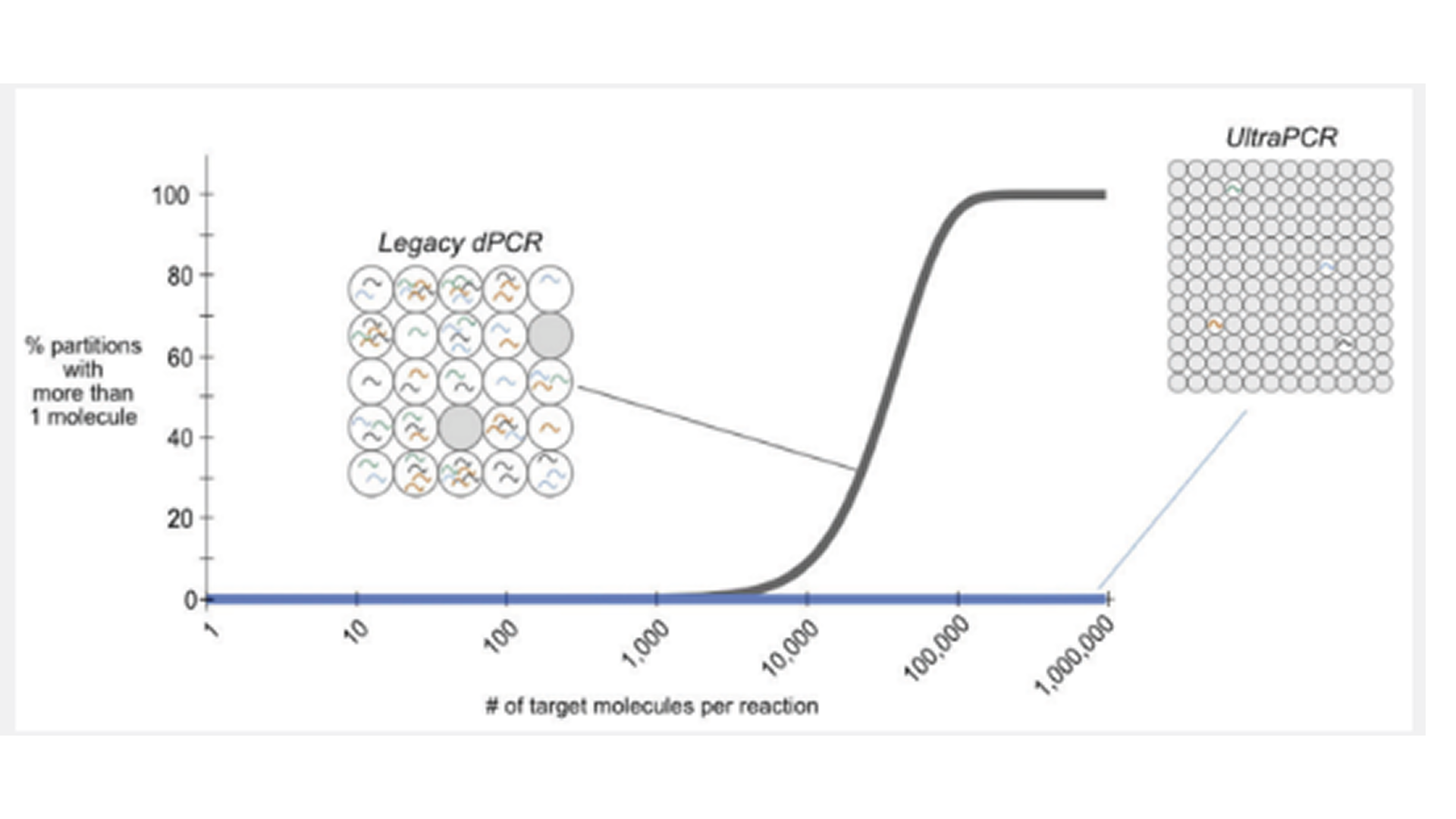
Rare molecule detection remains a challenge in liquid biopsy applications. Increasing assay sensitivity requires higher DNA input, yet conventional digital PCR (dPCR) systems are constrained by limited input capacity and partition number, restricting analytical range. We evaluated a next generation PCR platform, Countable PCR, which generates approximately 30 million partitions per 50 µL reaction and supports DNA inputs exceeding 1 µg, enabling “deep sampling” for improved rare variant detection. Using the clinically relevant KIT (D816V) mutation associated with systemic mastocytosis (SM) as a model, we analytically validated a multiplexed Countable PCR assay across 15 patient blood and bone-marrow samples at multiple testing sites and compared results with a reference dPCR assay. Standard dPCR achieved a 0.03% mutant allele frequency (MAF) limit of detection only by pooling three wells (78,000 partitions, 100 ng input), as higher inputs caused image saturation and loss of precision. In contrast, Countable PCR demonstrated accurate quantification from 150 to 1,452 ng DNA within a single well, maintaining linearity and low background. Across all samples, MAF values showed near-perfect concordance with the orthogonal method (Pearson coefficient = 0.9997; p = 1.03 × 10-22). Synthetic controls achieved 99.95% variant detection accuracy (5 variant-positive vs. 10,026 wild-type partitions), and contrived samples at 300 ng and 1 µg inputs exhibited 100% concordance across expected variant frequencies. These results demonstrate that Countable PCR enables high-input, single-molecule quantification with exceptional sensitivity and reproducibility, supporting streamlined, cost-efficient workflows for rare variant detection and precision molecular diagnostics in clinical laboratory settings.